Promising Biotech Ocular Gets Price Target Update After Groundbreaking Therapy News
Taking a closer look at Ocular Therapeutix after an intriguing trial that could introduce a pioneering drug to the U.S.
You've reached your free article limit
You've read 0 of 1 free Pro articles.
A biotech firm that many may not have heard of prior, Ocular Therapeutix (OCUL) , released data generated from a Phase 3 (late stage) trial of Axpaxil, which is the firm's experimental therapy for wet age-related macular degeneration (AMD).
After a loading phase, the trial compared a single dose of Axpaxil (0.45 miligrams) to a single dose of aflibercept (2 mg) in patriots suffering from wet AMD.
Superiority endpoints met with 74.1% of patients dosed with Axpaxil versus 56.6% of patients dosed with aflibercept. 65.9% of patients maintained vision at week 52 with Axpaxil versus 44.8% with patients dosed with aflibercept.
In addition, Axpaxil was generally well-tolerated throughout the trial with no treatment-related ocular serious adverse events (SAEs). The firm plans, subject to regulatory feedback, a new drug application (NDA) to the FDA for Axpaxil. If approved, this would be the first drug of its kind available to patients in the U.S.
Earnings and Fundamentals
Ocular released the firm's fourth quarter financial results almost two weeks ago. The firm posted a GAAP EPS of -$0.29 on revenue of $13.25 million. The top-line print disappointed, while showing a 22.4% year-over-year contraction, while the bottom-line actually beat Wall Street's expectations. However, expectations for the current quarter are for a nearly 17% increase in revenue even ahead of a potential new drug that would be in a class of its own for those in need.
For those who were about to ask, the firm has a cash position of $737.1 million on the books, no short-term debt and long-term debt of just $71.3 million. The cash burn over the trailing 12 months comes to $216.8 million, so these guys can afford to suffer just a bit longer and even ramp up costs related to the commercializing of the product.
My Thoughts
The stock is down 27% on the news and almost 61% since peaking back in December 2025. I will be accumulating this stock from here (the mid-$6s) down into the low $5s in speculation of an eventual rebound driven by actual sales. Overnight, analyst Serge Belanger (five stars out of five at TipRanks) of Needham reiterated both his "buy" rating on the stock as well as his $20 target price.
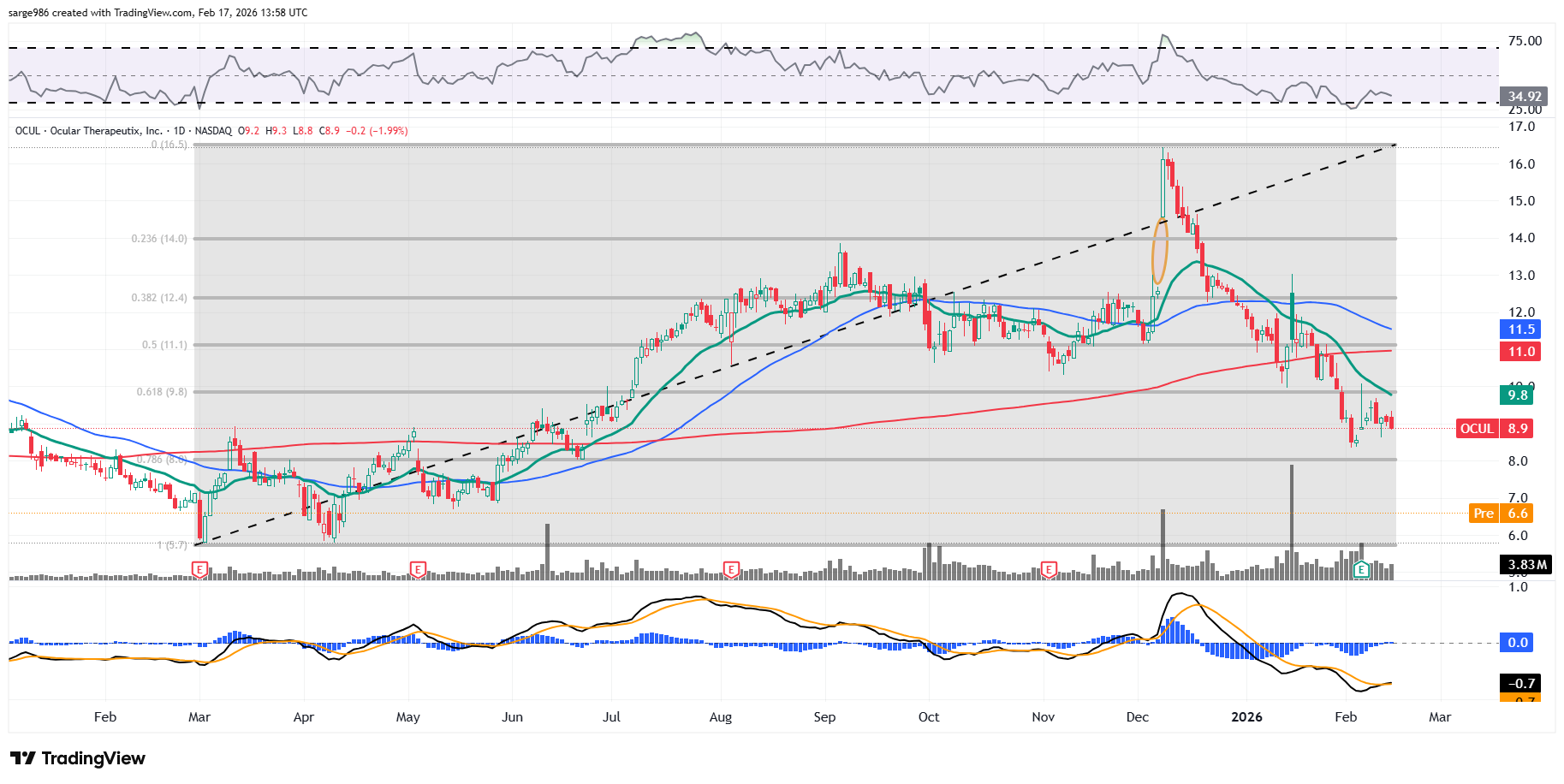
Readers will see that there had been an attempt to put in a bottom at the 78.6% Fibonacci retracement level of the March 2025 into December 2025 rally. That floor has cracked and my feeling is that, down to the 100% retracement level, the stock can be accumulated for the purpose of speculation.
I would not yet have a good target price as there is not yet a discernible pattern evident. That said, because I don't do anything without a target price or a panic point, I will be using the area in between the 50-day and 200-day SMAs and the loss of that March low.
At the time of publication, Guilfoyle had no positions in any securities mentioned.
