'Critical Treatment' Biotech Breaks Out on Real Profits and FDA Approval
This isn’t a 'hope and hype' biotech. Here's why the stock jumped and how to play it now.
You've reached your free article limit
You've read 0 of 1 free Pro articles.
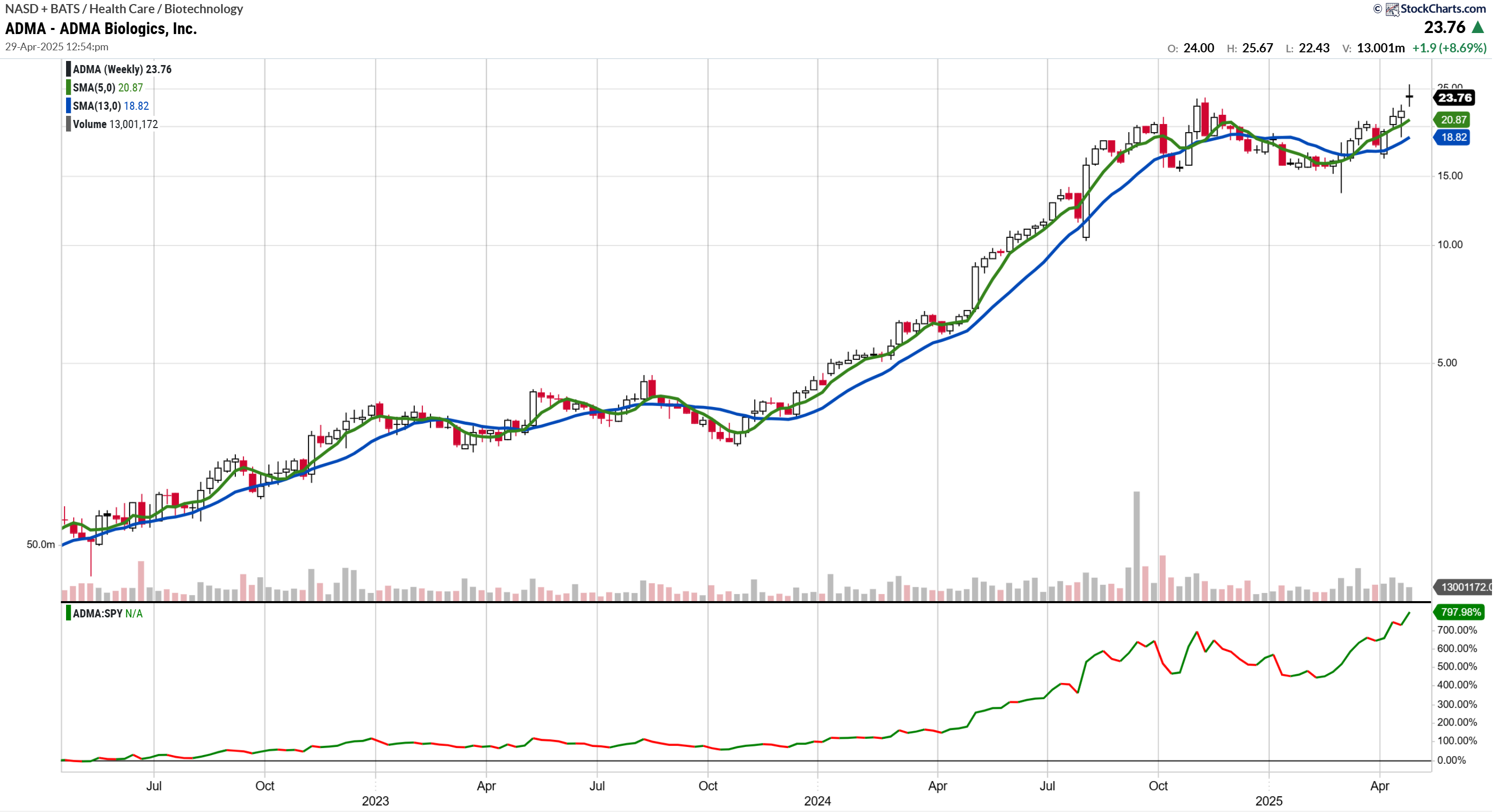
ADMA Biologics ADMA is shaping up to be one of the more interesting small-cap biotech stories out there right now.
It’s not a core holding, and it’s certainly not for clients looking for stability — but for those comfortable trading volatility with a fundamental tailwind behind it, this name deserves a closer look.
Why ADMA's Products Are Meaningful to Patients
ADMA Biologics makes plasma-based therapies that help people with weak immune systems — people who, without help, are walking around with no defense against common infections. Their main products, ASCENIV and BIVIGAM, are essentially giving these patients the antibodies they can’t produce on their own. It’s like borrowing someone else’s immune system when yours doesn’t show up to work.
This kind of treatment is critical. We're talking about people who would otherwise end up in the hospital (or worse) from things a healthy person shakes off in a day. So the demand for these therapies is consistent and growing — it’s not optional care, it’s essential.
Margins Are Likely to Expand Due to a New FDA Approved Method
ADMA just received FDA approval for a new process that boosts their production yield by about 20%. That means they can make a lot more medicine from the same amount of plasma — which is a big win, because plasma is scarce and expensive.
This process is designed to increase production yields by approximately 20% from the same starting plasma volume, benefiting both ASCENIV and BIVIGAM, two of the company's key immunoglobulin products,
In short: ADMA makes specialized medicines that help vulnerable people fight off disease. The new FDA approval lets them make more of it, faster and cheaper — which is good for patients and shareholders.
The Bull Case Is Getting Stronger
ADMA just posted record Q1 revenue — $81.9 million, up 44% from last year — and turned a profit for the first time. That’s a meaningful shift. They’re now projecting over $355 million in revenue for 2024 and north of $410 million next year. Management’s long-term target? Over $1 billion before 2030.
Margins are moving in the right direction too. Gross margin jumped from 29% to 48% year-over-year. They’re clearly gaining scale and finding some operating leverage in the model.
Product and Supply Chain Strength
This isn’t a “hope and hype” biotech. ASCENIV, their lead immunoglobulin therapy, is seeing real adoption, and they’ve secured high-titer plasma supply into the late 2030s. That locks in a critical input for long-term revenue durability. They’ve also submitted a manufacturing process update to the FDA to improve yields — something that could add more upside if approved.
The balance sheet’s no longer a liability either. Over $100 million in cash, net cash positive after paying down $30 million in debt. They’re positioned to reinvest without relying on dilution or junk financing.
But Make No Mistake — This Is a Trader’s Stock
Here’s the other side: ADMA is volatile. Price action can be erratic, and the stock is still small-cap biotech — thinly traded, sentiment-driven, and subject to headline risk.
Execution risk is still real. The business is heavily reliant on a single product line, and they’re competing with giants like Grifols and Takeda. One regulatory delay or supply chain hiccup can swing the story fast.
This is not a set-it-and-forget-it holding. This is for speculative capital, for traders who understand position sizing and risk management. The reward potential is there, but you need to respect the volatility.
How to Trade This Stock
That’s why the stock jumped. It’s not just a science win — it’s an operational and margin expansion win too. But like anything, there are risks: scaling up a new process isn’t always smooth, and if expectations get ahead of reality, the stock could pull back. Still, this is a meaningful step forward.
I took an initial position in the stock prior to the breakout on April 28, but given the potential upside is still in play, I would look to initiate a position on pullbacks. This is a very volatile stock so taking a smaller than normal position size is a reasonable strategy.
Bottom Line
ADMA is doing a lot of things right — growing revenue, scaling margins, managing capital responsibly. For speculative capital, it’s a name I’d keep on the screen. Just know what you’re holding: a company with big upside potential and the volatility to match.
At the time of publication, Llanes had a position in ADMA.
